| Cardiology Research, ISSN 1923-2829 print, 1923-2837 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, Cardiol Res and Elmer Press Inc |
| Journal website https://cr.elmerpub.com |
Original Article
Volume 17, Number 1, February 2026, pages 23-31
Assessment of No-Reflow in Patients With STEMI After Intracoronary Tirofiban After Opening of the Vessel
Mohammed Ali Mohammed Hammada , Wael Anwar Elshahat Hassiba
, Mohamed Kamal Ibrahim Salamaa
, Husna Irfan Thalibb, g
, Mohammed Moanesc, d, Muhammad Reihane, f
aCardiology Department, Faculty of Medicine, Kafr Elsheikh University, Kafr Elsheikh, Egypt
bGeneral Medicine Practice Program, Batterjee Medical College, Jeddah, Saudi Arabia
cCardiology Department, Faculty of Medicine, Al-Azhar University, Cairo, Egypt
dHayat National Hospitals, Saudi Arabia
eCardiology Department, Faculty of Medicine, Al-Azhar University, Damietta, Egypt
fDepartment of Internal Medicine, Batterjee Medical College, Jeddah 21442, Saudi Arabia
gCorresponding Author: Husna Irfan Thalib, General Medicine Practice Program, Batterjee Medical College, Jeddah, Saudi Arabia
Manuscript submitted December 22, 2025, accepted February 11, 2026, published online February 28, 2026
Short title: No-Reflow Assessment After IC Tirofiban in STEMI
doi: https://doi.org/10.14740/cr2180
| Abstract | ▴Top |
Background: No-reflow phenomenon (NRP) following primary percutaneous coronary intervention (PPCI) remains a critical determinant of adverse outcomes in ST-segment elevation myocardial infarction (STEMI) cases despite successful epicardial recanalization. The core purpose of this study was to establish the value of intracoronary (IC) tirofiban, delivered via the IC route, in mitigating the occurrence of NRP for STEMI cases subsequent to successful vessel reopening.
Methods: This randomized controlled double-blind study enrolled 60 STEMI cases. Following successful PCI, cases with thrombolysis in myocardial infarction (TIMI) flow grade less than 3 were randomized to receive either IC tirofiban (25 ug/kg) or saline 0.9% as placebo, in addition to standard pre-procedural therapy with aspirin, heparin, and ticagrelor. TIMI flow grade and incidence of NRP were evaluated. Additionally, ST-T normalization in electrocardiogram (ECG) was assessed. Bleeding complications and major adverse cardiac events (MACEs) were recorded during hospitalization and at 30-day follow-up.
Results: The tirofiban group demonstrated notably superior coronary flow restoration with 80% achieving TIMI 3 flow versus 46.67% in controls (P = 0.007). NRP occurred in 20% of tirofiban cases compared to 53.33% in controls (P = 0.007). Minor bleeding complications increased in the tirofiban group (26.67% versus 3.33%, P = 0.026), while major bleeding remained absent in both groups. Total in-hospital MACEs were notably reduced with tirofiban treatment compared to controls (3.33% versus 30%, P = 0.012).
Conclusions: In STEMI cases following PPCI, IC tirofiban administration effectively reduces NRP, improves coronary flow restoration, and reduces MACE despite increased minor bleeding risk.
Keywords: Glycoprotein IIb/IIIa inhibitor; Intracoronary; No-reflow phenomenon; Percutaneous coronary intervention; ST-segment elevation myocardial infarction; Tirofiban
| Introduction | ▴Top |
Acute ST-segment elevation myocardial infarction (STEMI), a critical cardiovascular emergency caused by thrombotic occlusion following plaque rupture or erosion, is best treated with primary percutaneous coronary intervention (PPCI), which offers superior outcomes over thrombolytic therapy [1]. Nevertheless, a sequence of harmful processes, known together as ischemia-reperfusion injury, is paradoxically set in motion by the re-establishment of coronary blood flow to formerly ischemic heart tissue [2].
The pathophysiological mechanisms underlying myocardial ischemia-reperfusion injury encompass a complex interplay of cellular death pathways, including apoptosis, necrosis, and autophagy, alongside maladaptive processes such as cardiomyocyte hypertrophy, interstitial fibrosis, and aberrant angiogenesis [3]. These molecular events manifest clinically as diverse phenomena such as lethal reperfusion trauma, microvascular impairment, myocardial dysfunction, and arrhythmias induced by reperfusion, all of which can substantially compromise patient outcomes despite successful epicardial recanalization [4].
Microvascular obstruction, a common and serious complication after coronary reperfusion for acute myocardial infarction, involves structural and functional damage to the coronary microvasculature, leading to a no-reflow phenomenon (NRP) despite successful epicardial reperfusion [5]. The NRP has been identified as one of the primary determinants of adverse cardiovascular outcomes in cases with STEMI, notably impacting short and long-term prognosis [6].
Successful recanalization of the infarct-related artery (IRA) with restoration of normal coronary flow is fundamentally associated with preserved left ventricular function and reduced mortality rates in cases with STEMI [7]. Conversely, the occurrence of NRP notably attenuates the clinical benefits derived from successful epicardial recanalization, highlighting the critical importance of maintaining optimal microvascular perfusion [8].
Contemporary therapeutic approaches have increasingly focused on both mechanical and pharmacological interventions to optimize myocardial reperfusion, with particular emphasis on the adjunctive use of glycoprotein IIb/IIIa inhibitors in cases undergoing PPCI [9]. While current clinical practice guidelines recommend the administration of small molecule glycoprotein IIb/IIIa inhibitors as upstream therapy followed by continuous infusion in selected STEMI cases, evolving clinical practices and newer antiplatelet strategies have influenced the utilization patterns of these agents [10]. Accumulated evidence from multiple clinical investigations has demonstrated that both intravenous (IV) and intracoronary (IC) administration of glycoprotein IIb/IIIa inhibitors can notably improve clinical outcomes and reduce mortality in cases with STEMI [11, 12].
Tirofiban represents a small molecule nonpeptide tyrosine derivative classified within the glycoprotein IIb/IIIa inhibitor family [13]. Regarded as the most potent available inhibitors of platelet aggregation, these agents operate by competitively preventing both fibrinogen and von Willebrand factor from attaching to the glycoprotein IIb/IIIa receptor complex located on the platelet surface [14].
The IC administration of tirofiban offers theoretical and practical advantages over systemic delivery by achieving higher local concentrations at the thrombus site, enhancing receptor occupancy and platelet inhibition, and thereby promoting more effective thrombus resolution [15].
Given these considerations, the present investigation seeks to evaluate the incidence and determinants of NRP in cases with STEMI following IC administration of glycoprotein IIb/IIIa inhibitors after successful epicardial vessel recanalization.
| Materials and Methods | ▴Top |
This randomized controlled double-blinded study was conducted on 60 individuals aged 18 years or older of both sexes presenting with STEMI at Kafr Elsheikh University Hospital, Egypt from May 2025 to October 2025.
A diagnosis of STEMI was established when cases presented with typical chest pain lasting in excess of 30 min, accompanied by either an ST-segment elevation surpassing 1 mm in at least two adjacent electrocardiographic leads or the acute onset of a left bundle branch block [16].
The study was approved by the institutional ethical committee (ID: KFSIRB200-587), registered on ClinicalTrials.gov (ID: NCT06966674). Informed written consent was obtained from all participants.
Exclusion criteria were prior thrombolytic therapy within 24 h, known malignancies, thrombocytopenia, end-stage liver disease, cardiogenic shock, renal failure with a glomerular filtration rate below 30 mL/min, or any contraindication to tirofiban administration.
Randomization and blindness
Random allocation was performed using an online randomization program [17] to generate the randomization sequence. Randomization was stratified considering age, sex, IRA, and comorbidities to ensure balanced clinical characteristics between groups. Individual patient codes were secured in opaque sealed envelopes to maintain allocation concealment. Participants were randomly assigned in a 1:1 ratio using parallel group allocation to one of two treatment arms. The study group (tirofiban group, n = 30) received IC tirofiban (Aggrastat®, Correvio LLC, USA) at a dose of 25 µg/kg, while the control group (placebo group, n = 30) received IC 0.9% saline solution as placebo. The selected dose of 25 µg/kg for IC tirofiban was based on prior studies demonstrating effective platelet inhibition with minimal systemic bleeding risk. A rigorous double-blinding methodology was implemented wherein both the cases and the outcome assessors remained unaware of group allocation throughout the study period. The interventional medications were prepared by a pharmacist who was not involved in subsequent patient management or data collection. The syringes were labeled only with the patient’s study number and were identical in appearance, volume, and color, thereby maintaining the integrity of the blinding process. Tirofiban was administered as a single IC bolus without subsequent prolonged IV infusion.
Comprehensive medical and surgical histories were obtained from all participants. Physical examinations were performed and routine laboratory investigations were conducted according to standard protocols prior to the intervention. An electrocardiogram (ECG) was done to assess the ST segment shift either elevation or depression, rhythm assessment, and T wave changes.
PPCI protocol
All cases received standardized pre-procedural medication involved a 300 mg oral dose of aspirin, IV heparin at 70 U/kg, and a 180 mg oral ticagrelor loading dose. Within the cardiology catheterization laboratory, all PPCI procedures were performed utilizing the femoral approach as the sole access site. In situations permitting, direct stenting constituted the chosen intervention technically feasible, with balloon pre-dilatation reserved for cases where direct stenting was not appropriate. The intervention was limited to the IRA only.
A residual stenosis of less than 50% and the presence of thrombolysis in myocardial infarction (TIMI) grade 3 flow post-intervention indicated procedural success. Conversely, NRP was characterized by post-procedural TIMI flow grades of 0, 1, or 2. All procedures used femoral access due to operator experience and catheterization lab constraints. No radial procedures were performed. All bleeding complications were categorized by access site; no non-access site major bleeding occurred.
Management of NRP and study intervention
Following stent implantation, cases demonstrating TIMI flow grade less than 3 underwent additional post-dilatation procedures with balloon catheters. IC vasodilator agents, including nitrates and diltiazem, were administered in clinically appropriate cases. Cases who continued to exhibit TIMI flow grade less than 3 despite these interventions were classified as having NRP and subsequently randomized to receive either the active treatment or placebo.
The study intervention involved administration of either IC tirofiban (25 µg/kg) or saline solution 0.9% as placebo through the guiding catheter into the IRA. For lesions located in the circumflex or left anterior descending arteries, the intervention was delivered from the left main coronary artery. To assess the acute effects of tirofiban on coronary NRP, the bolus dose was administered directly into the IC circulation following successful recanalization of the thrombotic occlusion.
Repeat coronary angiography was performed 10 min following the intervention, and TIMI flow grades were recorded for both treatment groups.
Outcome measures and assessment criteria
Coronary blood flow was assessed using TIMI flow grades and TIMI frame count before and within 15 min after treatment [18]. The TIMI bleeding criteria served as the basis for categorizing hemorrhagic complications, where major bleeding was identified by the occurrence of intracranial hemorrhage or a hemoglobin reduction surpassing 50 g/L, while minor bleeding encompassed visible bleeding events with a hemoglobin drop > 30 g/L. A platelet count less than 60 × 109/L was utilized to define thrombocytopenia.
The occurrence of major adverse cardiac events (MACEs) in addition to post-infarction angina and repeat revascularization was noted throughout hospitalization and again at the 30-day follow-up.
The primary outcome was coronary artery blood flow evaluated by TIMI flow grade and frame count pre- and post-treatment. TIMI flow grading was performed independently by two blinded interventional cardiologists, and disagreements were resolved by consensus. Secondary outcome was incidence of MACE within 30 days post-intervention. Left ventricular function was assessed by transthoracic echocardiography at baseline and at 30-day follow-up to monitor changes in ejection fraction and wall motion abnormalities.
Sample size calculation
G*Power 3.1.9.2 (Universitat Kiel, Germany) was employed for sample size estimation. Previous research [19] indicated mean ± SD IRA blood flow, as measured by the enhanced TIMI frame count method, to be 1.68 ± 0.23 mL/s in the tirofiban group and 1.42 ± 0.31 mL/s in the control group. The sample size was derived using an effect size of 0.953, a 95% confidence limit, 90% power for the study, and a 1:1 group ratio. To accommodate for potential participant attrition, five additional cases were incorporated into each group, leading to a planned recruitment of 30 cases per group.
Statistical analysis
SPSS v29 (IBM©, Armonk, NY, USA) was the software employed for all statistical procedures. The normality of data distribution was evaluated using the Shapiro-Wilk test in conjunction with histograms. Quantitative parametric data were presented as mean and SD and subsequently analyzed utilizing the unpaired Student’s t-test. Qualitative variables were detailed by frequency and percentage, with their analysis performed using the Chi-square test or Fisher’s exact test where applicable. A two-tailed P value ≤ 0.05 was pre-determined as the criterion for statistical significance.
| Results | ▴Top |
A total of 73 cases were screened for potential enrollment in the study. Of these, nine did not fulfill the specified criteria, and four elected not to participate. Consequently, the remaining 60 cases were randomly assigned to one of two groups, with each placebo group containing 30 cases. Comprehensive follow-up and statistical analysis were conducted for all allocated cases (Fig. 1).
 Click for large image | Figure 1. CONSORT flowchart of the enrolled patients. |
Age, sex, BMI, comorbidities, or laboratory parameters including hemoglobin, creatinine, liver enzymes, lipid profile, and troponin-I were similar across groups (Table 1).
 Click to view | Table 1. Pre-Interventional Data and Laboratory Tests |
No substantial differences were observed between the two groups in the IRA, balloon predilation, stent length, stent diameter, and stent inflation pressure. All cases in both groups needed stent implantation (Table 2).
 Click to view | Table 2. Interventional Data |
Pre-PCI TIMI flow was grade 0 in all cases. Post-tirofiban, TIMI flow grade, reflow, and ST-T normalization were notably higher in group S than group C (P = 0.041, 0.007, and 0.034). Treatment for no-reflow post-PCI (IC nitrate and diltiazem) was notably lower in group S (P = 0.007). Post-PCI balloon dilatation showed no substantial difference between groups. Minor bleeding was notably higher in group S (P = 0.026), while major bleeding did not occur in either group. Cardiac death was not notably different between groups. Total in-hospital target vessel failure (TVF) was notably lower in group S compared to group C (P = 0.012) (Table 3, Fig. 2). Representative angiographic images of the right coronary artery (RCA) before and after PCI, with and without IC tirofiban, are shown in Figure 3. Post-tirofiban administration, TIMI 3 flow was achieved, illustrating improved coronary perfusion compared to the placebo group.
 Click to view | Table 3. TIMI Flow Grade, ST-T Normalization, Treatment Outcomes, and Complications |
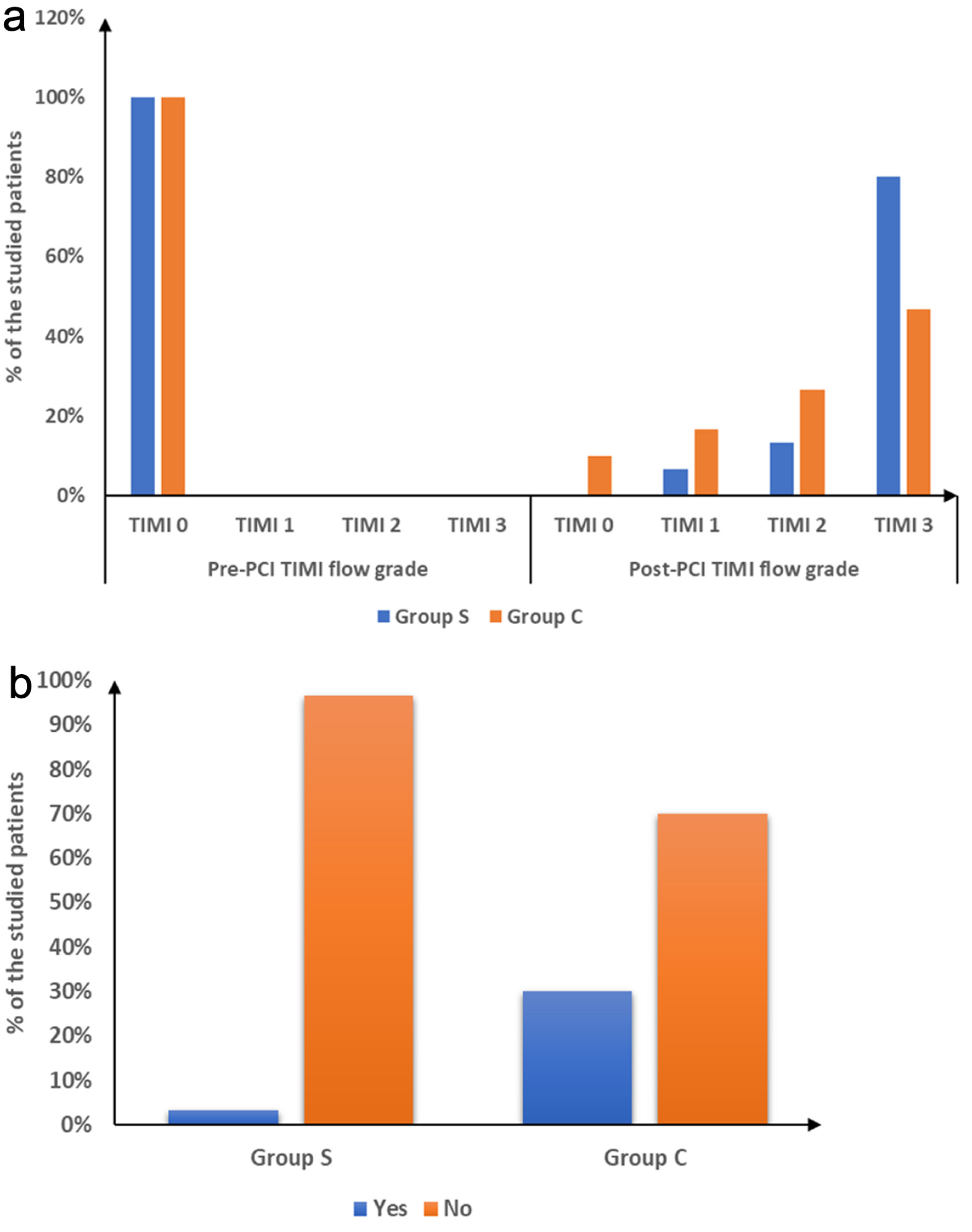 Click for large image | Figure 2. (a) Pre- and post-PCI TIMI flow grade and (b) total in hospital TVF of the studied groups. PCI: percutaneous coronary intervention; TIMI: thrombolysis in myocardial infarction; TVF: target vessel failure. |
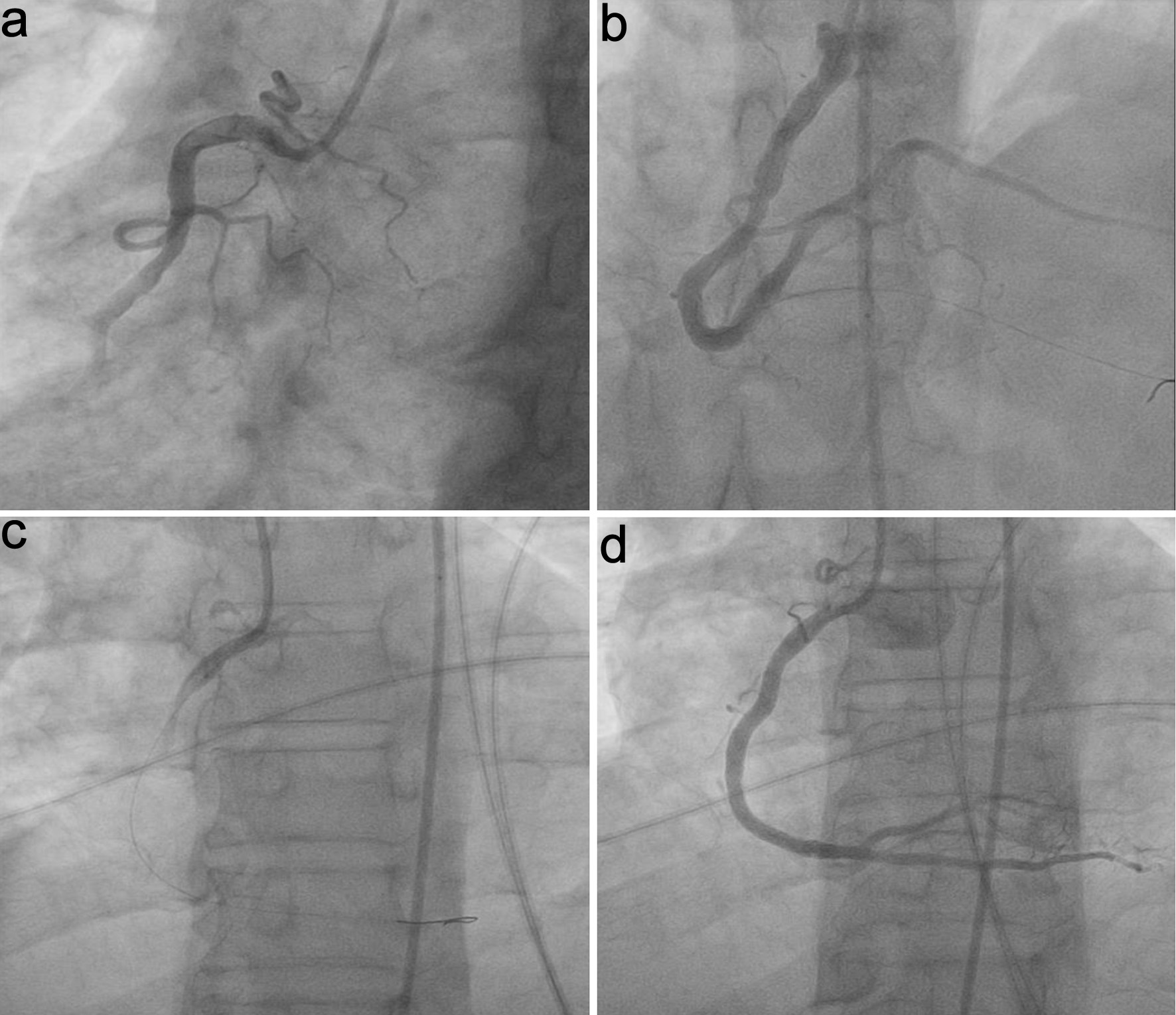 Click for large image | Figure 3. Representative angiographic images of the right coronary artery (RCA) in STEMI patients. (a) Pre-PCI, placebo group. (b) Post-PCI, placebo group. (c) Pre-PCI, IC tirofiban group. (d) Post-PCI, IC tirofiban group, demonstrating improved TIMI 3 flow with tirofiban administration. IC: intracoronary; PCI: percutaneous coronary intervention; TIMI: thrombolysis in myocardial infarction. |
| Discussion | ▴Top |
The management of STEMI through PPCI represents a critical therapeutic intervention, yet the occurrence of NRP continues to pose substantial challenges that adversely impact patient outcomes and long-term prognosis [20, 21].
This study shows that IC tirofiban notably improved coronary flow with higher TIMI 3 flow rates in STEMI cases, matching the meta-analysis by Shi et al [22], which demonstrated that IC low-dose tirofiban notably improved TIMI flow grade compared to IV administration (P = 0.037). Similarly, Akpek et al [16] reported successful reperfusion in 32% of cases receiving IC tirofiban compared to only 10% in the placebo group, with substantial improvement in TIMI flow grade (P < 0.001).
The mechanism underlying these improvements appears related to the direct local delivery of tirofiban to the site of coronary occlusion, allowing for higher local drug concentrations while minimizing systemic exposure [14]. Wang et al [19] corroborated these findings in cases with serious thrombus burden, reporting increased TIMI-3 flow in the IRA (89.3% versus 85.4%, P < 0.05) and enhanced blood flow calculated using TIMI frame count methodology. The superior efficacy of IC versus IV administration was further substantiated by Tang et al [23], who observed improved TIMI flow grading in the IC group (P = 0.022).
The substantial reduction in NRP incidence observed in our study represents a clinically substantial finding that corresponds with existing literature on IC tirofiban efficacy. This improvement directly translates to reduced need for rescue interventions, as evidenced by the decreased requirement for IC nitrate and diltiazem administration (20% versus 46.67% for both interventions). Duan et al [24] found that IC tirofiban notably improved coronary blood flow and myocardial perfusion after NRP, achieving TIMI grade 3 flow in 76.5% of cases versus 52.9% with nitroprusside (P = 0.03).
The temporal aspect of tirofiban administration appears crucial for optimal outcomes, as demonstrated by Mei and Yu [25] who found that timing of tirofiban administration is critical, with delivery during TIMI grade ≥ 1 yielding better blood flow and reduced myocardial injury compared to administration without forward flow. Gao et al [26] further supported these observations, reporting lower incidence of NRP (P = 0.031) when thrombectomy was combined with IC tirofiban administration compared to thrombectomy alone or neither treatment.
Although a lower in-hospital MACE rate was observed in the tirofiban group (3.33% vs. 30%), the study was not powered to detect differences in MACE; these findings should be interpreted as hypothesis-generating. Supporting evidence from Ma et al [27] using cardiac MRI showed that IC tirofiban reduced microvascular obstruction (56% versus 36%, P = 0.004), improved left ventricular strain (−12.5 versus −12.3, P = 0.042), and enhanced myocardial perfusion index (0.11 versus 0.09, P = 0.026), demonstrating superior cardiac outcomes compared to IV administration in cases with myocardial infarction.
The preservation of left ventricular function represents a critical long-term benefit, as demonstrated by Chen et al [28], who reported notably higher left ventricular ejection fraction and lower incidence of MACE in elderly STEMI cases receiving combined IC tirofiban and nicorandil [29]. Tian et al [12] conducted a comprehensive meta-analysis revealing that IC tirofiban notably reduced 30-day MACE (P = 0.028) and improved both in-hospital and 6-month left ventricular ejection fraction compared to IV administration.
The observed increase in minor bleeding complications (26.67% versus 3.33%) in our study population warrants careful consideration within the context of overall risk-benefit assessment. Despite this increase in minor bleeding events, the absence of major bleeding complications in both groups suggests an acceptable safety profile for IC tirofiban administration. This safety pattern aligns with findings from Shi et al [22] which revealed no substantial variation in the incidence of bleeding events between IC and IV tirofiban administration in their meta-analysis.
The bleeding risk associated with IC tirofiban administration must be contextualized against the substantial reduction in MACE and improved coronary perfusion outcomes. Qin et al [20] acknowledged a trend toward increased bleeding risk in their meta-analysis but noted that the data did not reach statistical significance. Similarly, Tian et al [12] reported no substantial difference in in-hospital bleeding or thrombocytopenia between IC and IV tirofiban, supporting its acceptable safety profile alongside clinical benefits.
While IC tirofiban has been previously studied, our investigation confirms its efficacy in a contemporary STEMI cohort and serves as an angiographic proof-of-concept; we acknowledge that novelty is limited. Our study has several limitations. TIMI flow grades and TIMI frame count, although commonly used, have low specificity for detecting microvascular obstruction. This is a single-center study with a small sample size and short follow-up, which may limit generalizability. The 30-day follow-up precludes assessment of long-term outcomes. Reliance on TIMI flow grades may overlook nuanced microvascular dysfunction. Lastly, we did not assess left ventricular function with cardiac MRI or directly measure infarct size, which could provide more precise information on myocardial salvage. Despite observed benefits, IC tirofiban is not guideline-endorsed due to limited large-scale randomized trials, heterogeneity of study designs, and short follow-up in existing literature.
Conclusions
IC tirofiban (25 µg/kg) notably reduced NRP incidence and improved TIMI 3 flow restoration versus placebo in STEMI cases post-PCI. It lowered in-hospital MACE despite increased minor bleeding. Tirofiban improved TIMI flow, which may indirectly reflect enhanced microvascular perfusion and contributes to improved short-term outcomes after epicardial recanalization, although direct measurement of microvascular function was not performed.
Acknowledgments
None to declare.
Financial Disclosure
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of Interest
The authors declare no conflict of interest related to this study.
Informed Consent
Informed written consent was obtained from all participants prior to enrollment in the study.
Author Contributions
Mohammed Ali Mohammed Hammad, Wael Anwar Elshahat Hassib, and Mohamed Kamal Ibrahim Salama: Conceptualization and methodology, patient recruitment, and data collection. Husna Irfan Thalib: Drafting of the manuscript, statistical analysis, and literature review. Mohammed Moanes Mohammed Mohyeldin and Muhammad Saad Reihan: Supervision, critical revision of the manuscript, and final approval of the version to be published. All authors have read and approved the final manuscript and agree to be accountable for all aspects of the work.
Data Availability
The data supporting the findings of this study are available from the corresponding author upon reasonable request.
| References | ▴Top |
- Ibanez B, James S, Agewall S, Antunes MJ, Bucciarelli-Ducci C, Bueno H, Caforio ALP, et al. 2017 ESC Guidelines for the management of acute myocardial infarction in patients presenting with ST-segment elevation: The Task Force for the management of acute myocardial infarction in patients presenting with ST-segment elevation of the European Society of Cardiology (ESC). Eur Heart J. 2018;39(2):119-177.
doi pubmed - Heusch G. Coronary microvascular obstruction: the new frontier in cardioprotection. Basic Res Cardiol. 2019;114(6):45.
doi pubmed - Niccoli G, Montone RA, Ibanez B, Thiele H, Crea F, Heusch G, Bulluck H, et al. Optimized treatment of ST-elevation myocardial infarction. Circ Res. 2019;125(2):245-258.
doi pubmed - Kleinbongard P, Heusch G. A fresh look at coronary microembolization. Nat Rev Cardiol. 2022;19(4):265-280.
doi pubmed - Ghobrial M, Bawamia B, Cartlidge T, Spyridopoulos I, Kunadian V, Zaman A, Egred M, et al. Microvascular obstruction in acute myocardial infarction, a potential therapeutic target. J Clin Med. 2023;12(18):5934.
doi pubmed - Hennawy BS, Mohammed A, Fouad KA. The relationship between high-dose atorvastatin loading and the no-reflow phenomenon in STEMI patients undergoing primary PCI. Iran Heart J. 2025;26:54-66.
- Samir A, Nagy S, Abdelhamid M, Kandil H. Clinical, electrocardiographic, echocardiographic, and angiographic predictors for the final infarct size assessed by cardiac magnetic resonance in acute STEMI patients after primary percutaneous coronary intervention. Egypt Heart J. 2024;76(1):111.
doi pubmed - Pelliccia F, Niccoli G, Zimarino M, Ando G, Porto I, Calabro P, De Rosa S, et al. Pathophysiology and treatment of the no-reflow phenomenon in ST-segment elevation myocardial infarction: focus on low-dose fibrinolysis during primary percutaneous intervention. Rev Cardiovasc Med. 2023;24(12):365.
doi pubmed - Saleiro C, Teixeira R, De Campos D, Lopes J, Oliveiros B, Costa M, Goncalves L. Glycoprotein IIb/IIIa inhibitors for cardiogenic shock complicating acute myocardial infarction: a systematic review, meta-analysis, and meta-regression. J Intensive Care. 2020;8(1):85.
doi pubmed - Hermanides RS, Kilic S, van 't Hof AWJ. Optimal pharmacological therapy in ST-elevation myocardial infarction-a review: a review of antithrombotic therapies in STEMI. Neth Heart J. 2018;26(6):296-310.
doi pubmed - El-Hefny E, Yassen I, M Osman M. Comparison between intracoronary versus intravenous bolus injection of tirofiban on infarct size during primary percutaneous coronary intervention in patients with acute anterior ST segment elevation myocardial infarction. Al-Azhar Med J. 2020;49:1313-1326.
- Tian R, Liu R, Zhang J, Li Y, Wei S, Xu F, Li X, et al. Efficacy and safety of intracoronary versus intravenous tirofiban in patients with ST-segment elevation myocardial infarction undergoing primary percutaneous coronary intervention: A meta-analysis of randomized controlled trials. Heliyon. 2023;9(5):e15842.
doi pubmed - Yang M, Huo X, Miao Z, Wang Y. Platelet glycoprotein IIb/IIIa receptor inhibitor tirofiban in acute ischemic stroke. Drugs. 2019;79(5):515-529.
doi pubmed - De Luca G, Verburg A, Hof AV, Ten Berg J, Kereiakes DJ, Coller BS, Gibson CM. Current and future roles of glycoprotein iib-iiia inhibitors in primary angioplasty for ST-segment elevation myocardial infarction. Biomedicines. 2024;12(9):2023.
doi pubmed - Wilmer CI. Intracoronary high-dose bolus tirofiban administration during complex coronary interventions: A United States-based case series. Cardiovasc Revasc Med. 2018;19(1 Pt B):112-116.
doi pubmed - Akpek M, Sahin O, Sarli B, Baktir AO, Saglam H, Urkmez S, Ergin A, et al. Acute effects of intracoronary tirofiban on no-reflow phenomena in patients with ST-segment elevated myocardial infarction undergoing primary percutaneous coronary intervention. Angiology. 2015;66(6):560-567.
doi pubmed - http://www.randomizer.org.
- Hong YJ, Jeong MH, Choi YH, Ko JS, Lee MG, Kang WY, Lee SE, et al. Predictors of no-reflow after percutaneous coronary intervention for culprit lesion with plaque rupture in infarct-related artery in patients with acute myocardial infarction. J Cardiol. 2009;54(1):36-44.
doi pubmed - Wang HL, Xing SY, Dong PS, Han YH, Zhu JH, Lai LH, Zhao JF. Safety and efficacy of intracoronary tirofiban administration in patients with serious thrombus burden and ST-elevation myocardial infarction undergoing percutaneous coronary intervention. Eur Rev Med Pharmacol Sci. 2014;18(23):3690-3695.
pubmed - Qin T, Xie L, Chen MH. Meta-analysis of randomized controlled trials on the efficacy and safety of intracoronary administration of tirofiban for no-reflow phenomenon. BMC Cardiovasc Disord. 2013;13:68.
doi pubmed - Ikramullah Hamidullah, Ahmad F, Khan S, Bilal HA, Samiurehman. Frequency of No-Reflow in Patients Undergoing Primary Percutaneous Coronary Intervention and the Impact of Intracoronary Adrenaline and Tirofiban on Timi Flow. J Ayub Med Coll Abbottabad. 2024;36(3):510-513.
doi pubmed - Shi L, Chen L, Tian W, Zhao S. Intracoronary versus intravenous low-dose tirofiban in patients with ST-elevation myocardial infarction: a meta-analysis of randomised controlled trials. Heart Lung Circ. 2024;33(11):1533-1542.
doi pubmed - Tang X, Li R, Zhang T. Comparison of intracoronary versus intravenous tirofiban in acute ST-elevation myocardial infarction patients undergoing primary percutaneous coronary intervention. Coron Artery Dis. 2022;33(7):547-552.
doi pubmed - Duan H, Wan X, Li B. Effects of intracoronary arterial injection of tirofiban on no-reflow phenomenon in patients with acute ST-segment elevation myocardial infarction underwent primary percutaneous coronary intervention. Discuss Clin Cases. 2017;4:1-5.
- Mei C, Yu H. Effectiveness of different tirofiban administration times in patients with no-reflow myocardial infarction during percutaneous coronary intervention. Pak J Med Sci. 2024;40(9):1969-1974.
doi pubmed - Gao L, Cao Z, Zhang H. Efficacy and safety of thrombectomy combined with intracoronary administration of tirofiban in ST-segment elevation myocardial infarction (STEMI). Med Sci Monit. 2016;22:2699-2705.
doi pubmed - Ma Q, Ma Y, Wang X, Li S, Yu T, Duan W, Wu J, et al. Intracoronary compared with intravenous bolus tirofiban on the microvascular obstruction in patients with STEMI undergoing PCI: a cardiac MR study. Int J Cardiovasc Imaging. 2020;36(6):1121-1132.
doi pubmed - Chen GX, Wang HN, Zou JL, Yuan XX. Effects of intracoronary injection of nicorandil and tirofiban on myocardial perfusion and short-term prognosis in elderly patients with acute ST-segment elevation myocardial infarction after emergency PCI. World J Emerg Med. 2020;11(3):157-163.
doi pubmed - Heusch G. Coronary blood flow in heart failure: cause, consequence and bystander. Basic Res Cardiol. 2022;117(1):1.
doi pubmed
This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (CC BY 4.0), which permits unrestricted use, distribution, and reproduction in any medium, including commercial use, provided the original work is properly cited.
Cardiology Research is published by Elmer Press Inc.