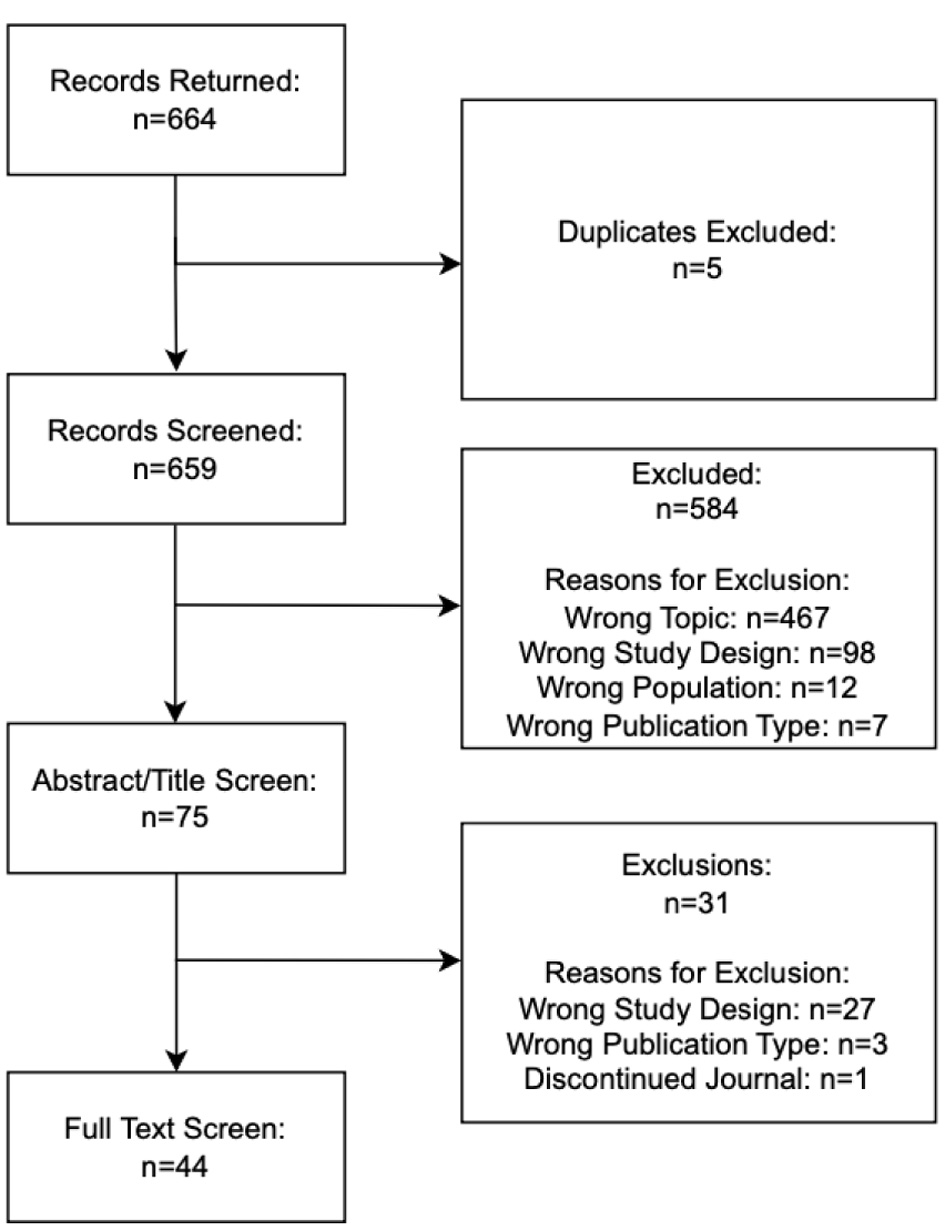
↓ Figure 1. PRISMA flow diagram.
| Cardiology Research, ISSN 1923-2829 print, 1923-2837 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, Cardiol Res and Elmer Press Inc |
| Journal website https://cr.elmerpub.com |
Original Article
Volume 17, Number 2, April 2026, pages 72-81
Integrating Findings Into Practice: Assessing External Validity of Congestive Heart Failure Trials
Figures

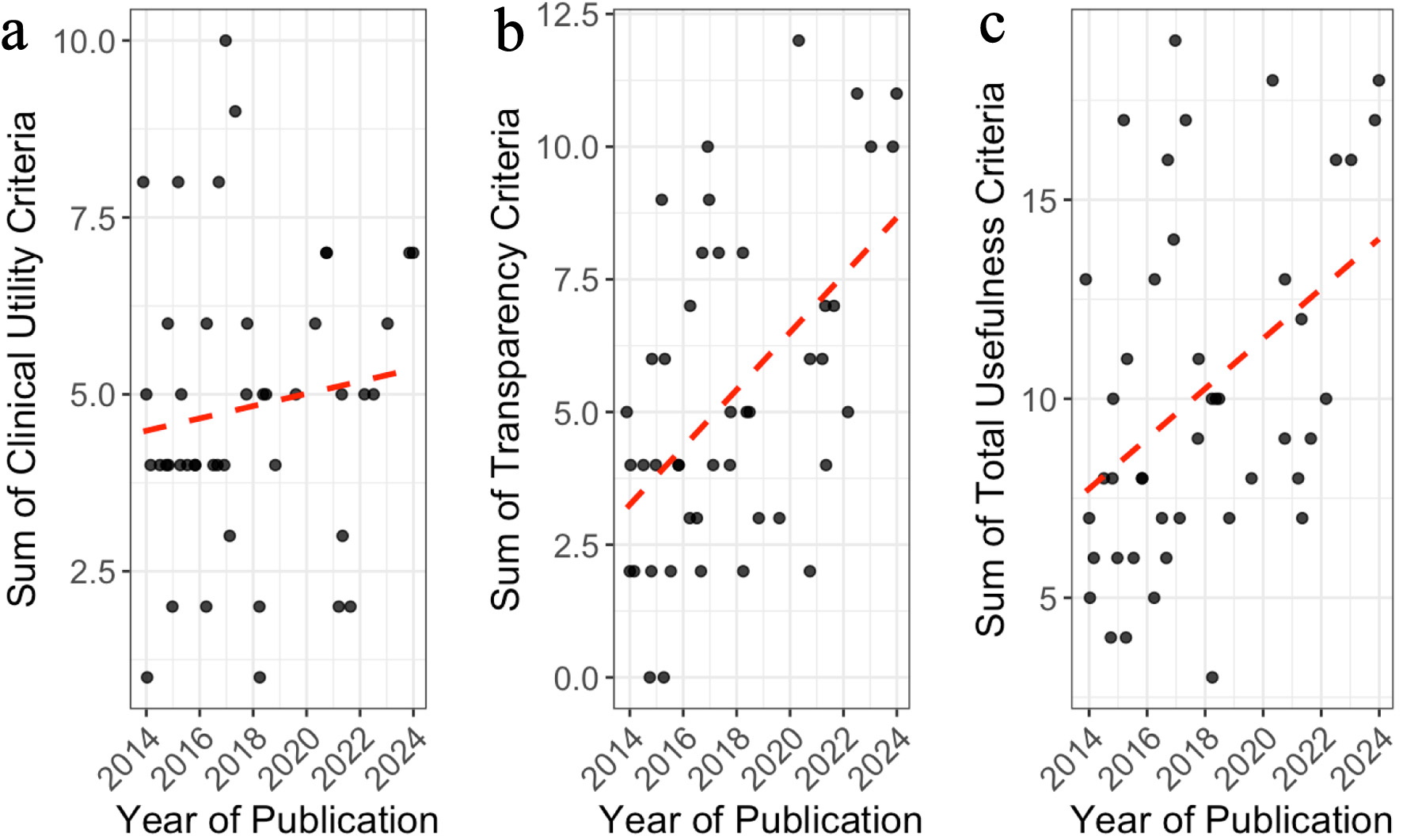
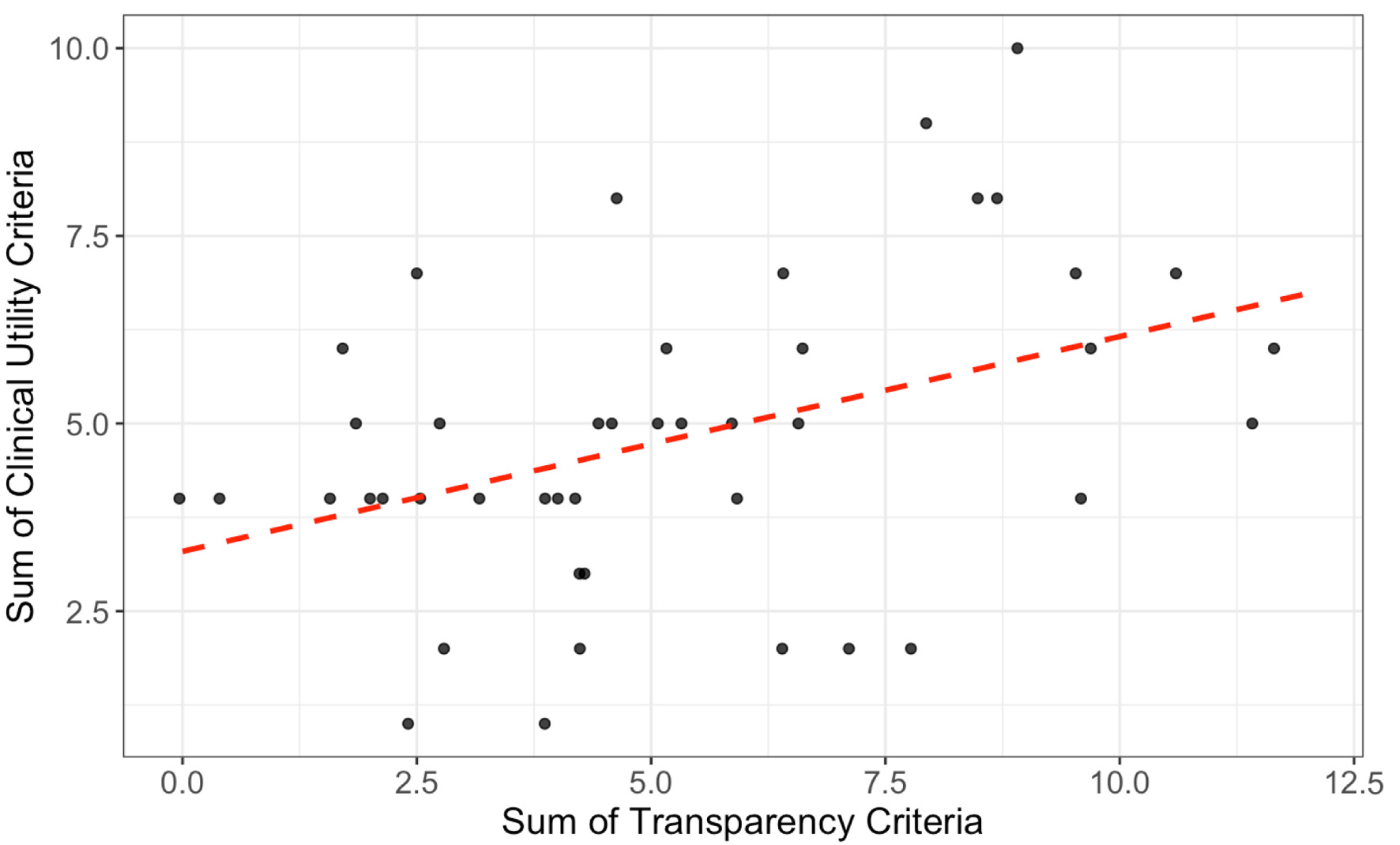
Tables
| Clinical utility item | Description |
|---|---|
| 1. Problem base | Health impact depends on prevalence, individual burden, and cost, including common and rare diseases. |
| 2. Context placement | Clinical research is most useful when informed by systematic reviews that integrate new findings with existing evidence. |
| 3. Information gain | Informative clinical research requires adequately powered studies focused on critical outcomes, avoiding surrogate measures, and integrating results with prior evidence for meaningful practice changes. |
| 4. Pragmatism | Pragmatic research evaluates the real-world effectiveness of interventions, emphasizing generalizability and practicality over controlled, ideal conditions. |
| 5. Patient-centeredness | Patient-centered research prioritizes questions and outcomes that matter most to patients, involving them in setting priorities and developing core outcomes. |
| 6. Value for money | Value-for-money research evaluates the expected benefits of a study against its costs, using analyses like value-of-information and budget impact to guide design and funding decisions. |
| 7. Feasibility | Feasibility in research depends on realistic recruitment goals, adequate power, and collaborative networks to overcome challenges like patient availability and overestimated sample sizes. |
| Transparency item | Description |
| 8A. Preregistration | Preregistration is the process of registering a study in an official registry database before recruiting the first patient. |
| 8B. Public protocol | The protocol and data analysis plan should be publicly available before the trial starts and before data analysis. |
| 8C. Protocol adherence | Any deviations from the original protocol should be justified, updated in the protocol, and reapproved by the ethics committee, with modification statements included in the manuscript. |
| 8D. Funding statement | Funding sources for trial conduction should be clearly stated. |
| 8E. Conflict of interest statement | Conflicts of interest should be clearly and completely disclosed. |
| 8F. Data availability | Freely available raw data, including statistical code and output, is becoming the norm to ensure transparency, facilitate meta-analyses, and address concerns about study trustworthiness. |
| Clinical utility criteria | N = 44 | Transparency criteria | N = 44 |
|---|---|---|---|
| Problem base, n (%) | Preregistration, n (%) | ||
| Full | 4 (9.1) | Full | 18 (40.9) |
| Partial | 25 (56.8) | Partial | 9 (20.5) |
| Absent | 15 (34.1) | Absent | 17 (38.6) |
| Context placement, n (%) | Public protocol, n (%) | ||
| Full | 16 (36.4) | Full | 6 (13.6) |
| Partial | 28 (63.6) | Partial | 7 (15.9) |
| Absent | 0 (0.0) | Absent | 31 (70.5) |
| Information gain, n (%) | Adherence to protocol, n (%) | ||
| Full | 7 (15.9) | Full | 12 (27.3) |
| Partial | 4 (9.1) | Partial | 0 (0.0) |
| Absent | 33 (75.0) | Absent | 32 (72.7) |
| Pragmatism, n (%) | Funding stated, n (%) | ||
| Full | 2 (4.5) | Full | 34 (77.3) |
| Partial | 3 (6.8) | Partial | 0 (0.0) |
| Absent | 39 (88.6) | Absent | 10 (22.7) |
| Patient centeredness, n (%) | COIs, n (%) | ||
| Full | 24 (54.5) | Full | 30 (68.2) |
| Partial | 6 (13.6) | Partial | 2 (4.5) |
| Absent | 14 (31.8) | Absent | 12 (27.3) |
| Value for money, n (%) | Raw data, n (%) | ||
| Full | 1 (2.3) | Full | 6 (13.6) |
| Partial | 1 (2.3) | Partial | 4 (9.1) |
| Absent | 42 (95.5) | Absent | 34 (77.3) |
| Feasibility, n (%) | |||
| Full | 17 (38.6) | ||
| Partial | 3 (6.8) | ||
| Absent | 24 (54.5) |
| Characteristic | N = 44 |
|---|---|
| aMultiple studies had more than one pragmatism violation, resulting in a total N = 65 for this section. | |
| Prior systematic review, n (%) | |
| Not cited or performed | 26 (59.1) |
| Systematic review cited | 18 (40.9) |
| Non-feasibility reason, n (%) | |
| No reason provided | 20 (45.5) |
| Study was feasible | 18 (40.9) |
| Attrition | 3 (6.8) |
| Low recruitment speed | 3 (6.8) |
| Pragmatism violationa, n (%) | |
| Blinding of assessors | 22 (50.0) |
| Placebo-controlled | 18 (40.9) |
| Employs a new intervention | 12 (27.3) |
| Single-centered trial | 9 (20.5) |
| No pragmatism violation | 3 (6.8) |
| Employs a new indication | 1 (2.3) |