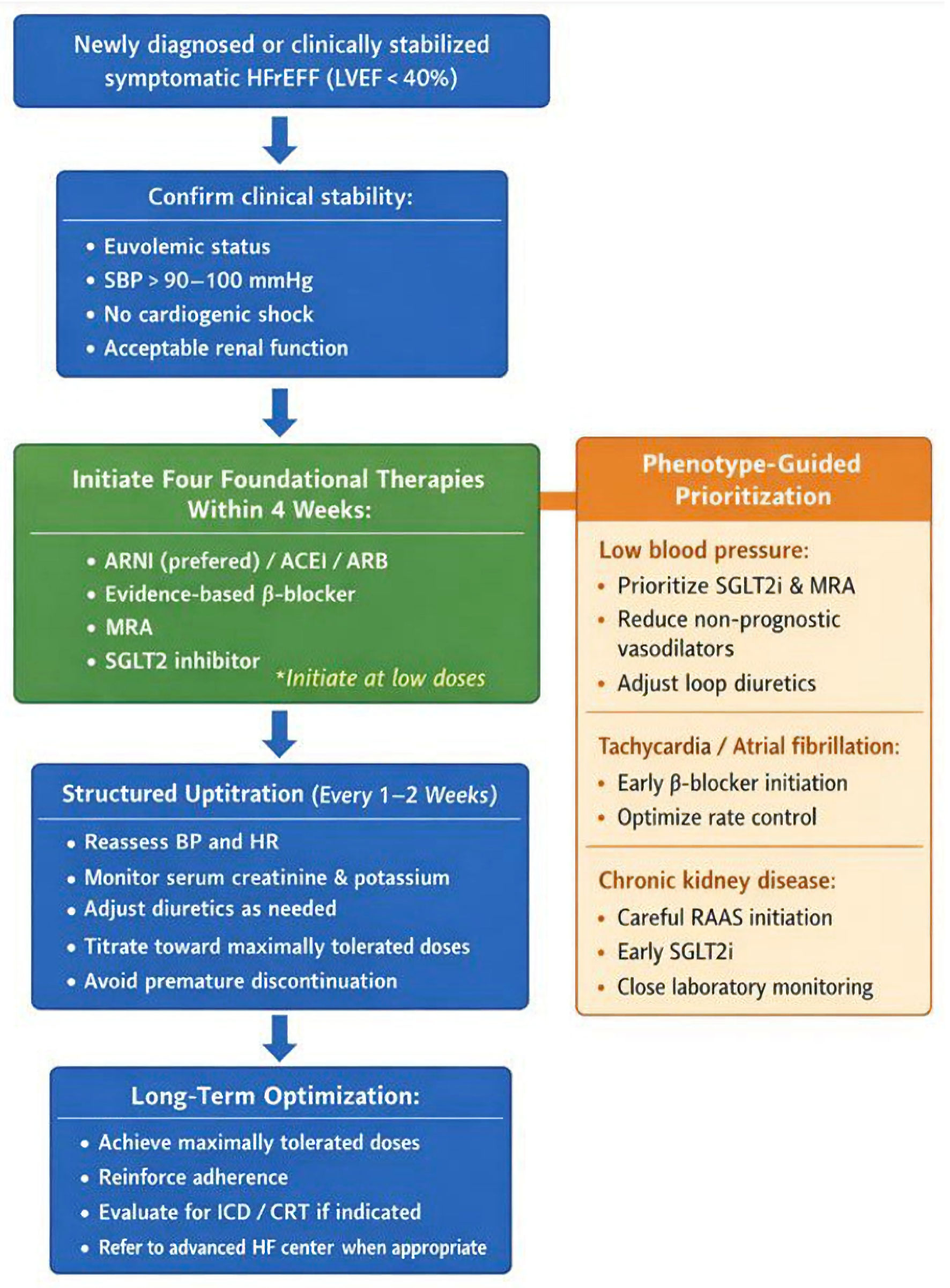
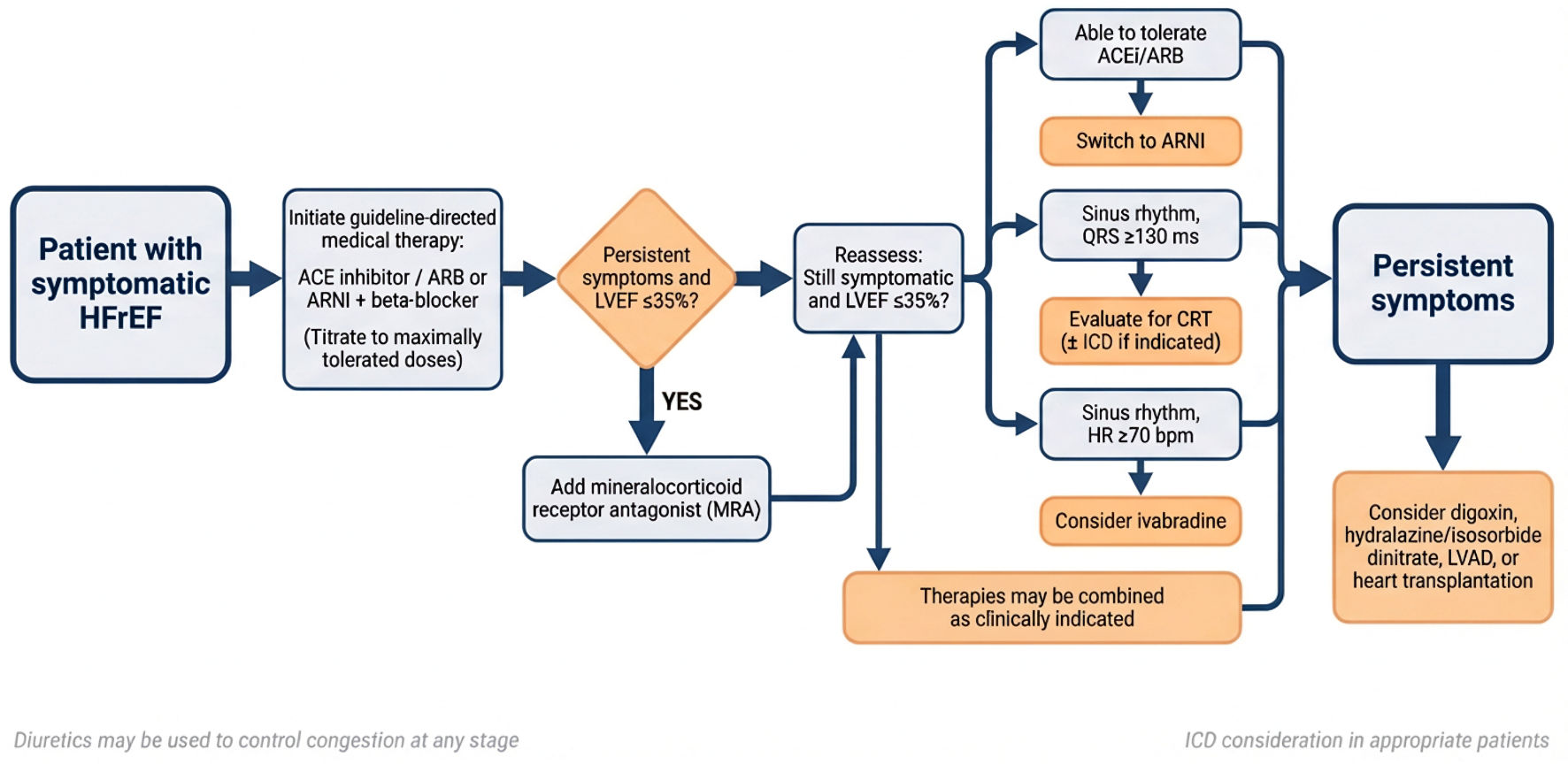
↓ Figure 1. Practical phenotype-guided framework for rapid initiation and optimization of guideline-directed medical therapy (GDMT) in patients with heart failure with reduced ejection fraction (HFrEF). The algorithm emphasizes early initiation of all four foundational therapies (“four drugs in 4 weeks”) followed by structured up-titration and phenotype-specific prioritization. ARNI: angiotensin receptor–neprilysin inhibitor; ACEi: angiotensin-converting enzyme inhibitor; ARB: angiotensin receptor blocker; MRA: mineralocorticoid receptor antagonist; SGLT2i: sodium–glucose cotransporter 2 inhibitor; BP: blood pressure; HR: heart rate; ICD: implantable cardioverter-defibrillator; CRT: cardiac resynchronization therapy; LVEF: left ventricular ejection fraction; SBP: systolic blood pressure.