Nuclear Respiratory Factor-1 Ameliorates Heart Failure by Suppressing Cardiomyocyte Pyroptosis-Associated Signaling Via the Downregulation of Gasdermin D and Caspase-1
DOI:
https://doi.org/10.14740/cr2153Keywords:
Heart failure, Pyroptosis, NRF-1, GSDMD, Caspase-1Abstract
Background: Cardiac diseases caused by various factors eventually lead to heart failure (HF) as the condition progresses, during which inflammation and pyroptosis are markedly enhanced. Nuclear respiratory factor-1 (NRF-1) is a transcriptional regulator involved in multiple physiological functions; however, its role in pyroptosis during HF remains unclear.
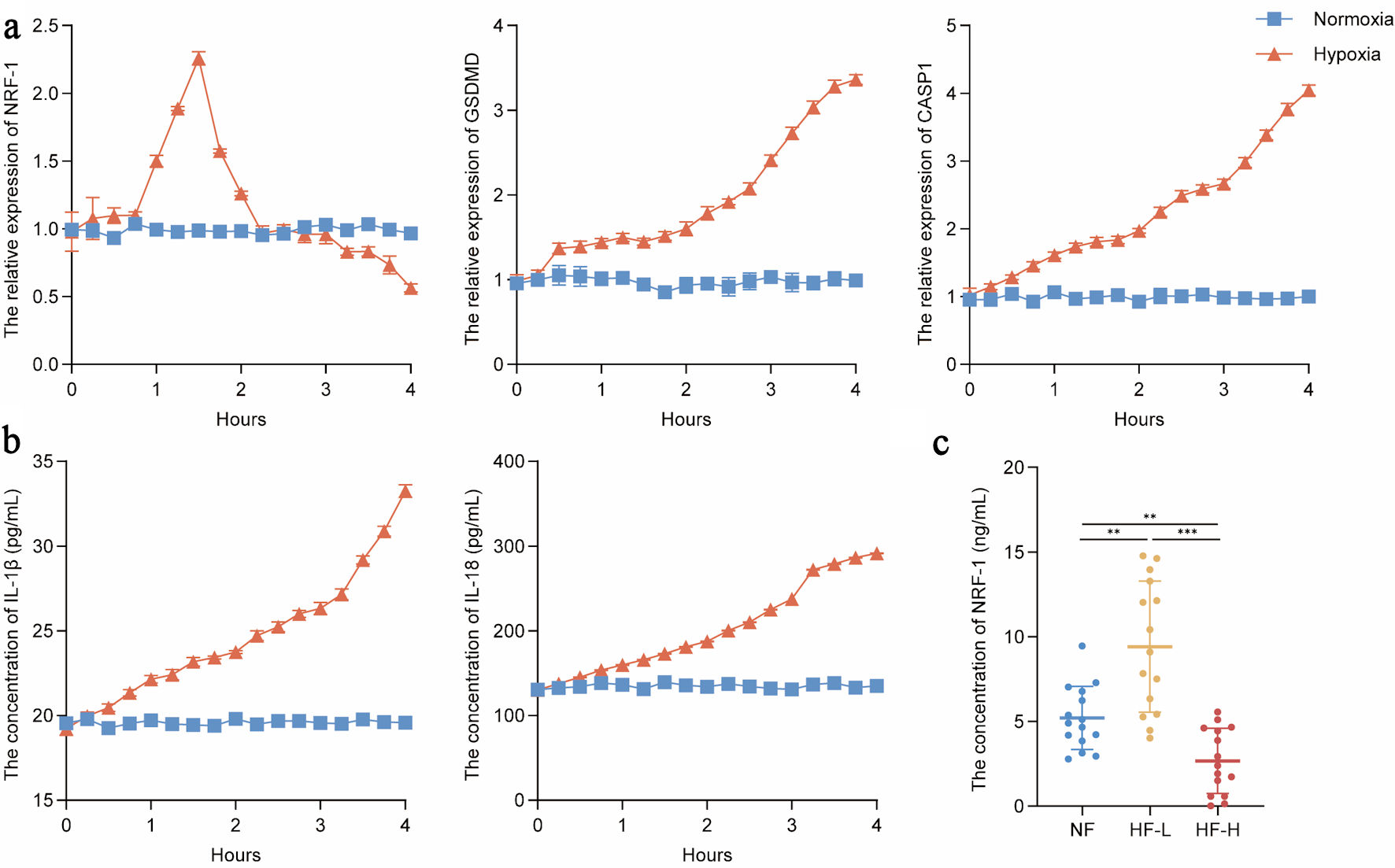
Methods: Serum samples from patients with HF were collected to evaluate the levels of NRF-1. An HF rat model was established to assess the expression of NRF-1 in serum and cardiac tissue and to investigate its association with HF and the expression of inflammatory markers gasdermin D (GSDMD), caspase-1, interleukin (IL)-8, and IL-1β. NRF-1-overexpressing and NRF-1-silenced H9C2 cell lines were constructed, and myocardial injury was induced by hypoxia and doxorubicin (DOX) to evaluate the effects of NRF-1 on pyroptosis-related molecules GSDMD and caspase-1, as well as inflammatory cytokines IL-8 and IL-1β. Finally, the expression of NRF-1 in the serum of HF patients was analyzed based on New York Heart Association (NYHA) functional classification to validate the dynamic changes of NRF-1 during pyroptosis in HF.
Results: Although previous studies have reported inconsistent findings regarding serum NRF-1 expression levels among different HF patient cohorts, our current results demonstrate that serum NRF-1 expression is significantly reduced in HF patients compared to those with normal cardiac function (NF), while the expression of pyroptosis-related molecules GSDMD and caspase-1, as well as pro-inflammatory cytokines IL-8 and IL-1β, is markedly increased. These findings were further validated in an HF rat model. In vitro experiments revealed that NRF-1 attenuates hypoxia and DOX-induced pyroptosis in H9C2 cardiomyocytes, highlighting its protective role in the pathogenesis of HF. Finally, serum NRF-1 levels assessed according to NYHA functional classification suggest that the differential expression of NRF-1 observed across samples may be attributed to variations in the stages of HF among patients.
Conclusions: NRF-1 is a dynamically expressed molecule with cardioprotective properties that ameliorates HF and attenuates pyroptosis by inhibiting the caspase-1/GSDMD signaling pathway.
Published
Issue
Section
License
Copyright (c) 2026 The authors

This work is licensed under a Creative Commons Attribution 4.0 International License.









