Integrating Findings Into Practice: Assessing External Validity of Congestive Heart Failure Trials
DOI:
https://doi.org/10.14740/cr2191Keywords:
Congestive heart failure, Randomized controlled trial, Patient-centered outcome, Pragmatic clinical trials, Research transparency, External validityAbstract
Background: Congestive heart failure (CHF) remains a major global health issue, affecting millions of adults worldwide, contributing to significant hospitalizations and mortality. While randomized controlled trials (RCTs) are essential for improving CHF care, their external validity remains uncertain. This study evaluates the external validity of CHF RCTs published between 2014 and 2024 using the criteria developed by van ’t Hooft et al.
Methods: A systematic appraisal of CHF RCTs was performed using MEDLINE and Embase on April 1, 2025. Included studies were full-text, English-language, human trials focused on CHF interventions. Trials were assessed with a 13-criterion guideline covering pragmatic principles, context, information gain, feasibility, transparency, value, and patient-centeredness. Two reviewers independently screened and extracted data, resolving discrepancies by consensus. Trial characteristics and predictors of usefulness were analyzed via linear regression and descriptive statistics.
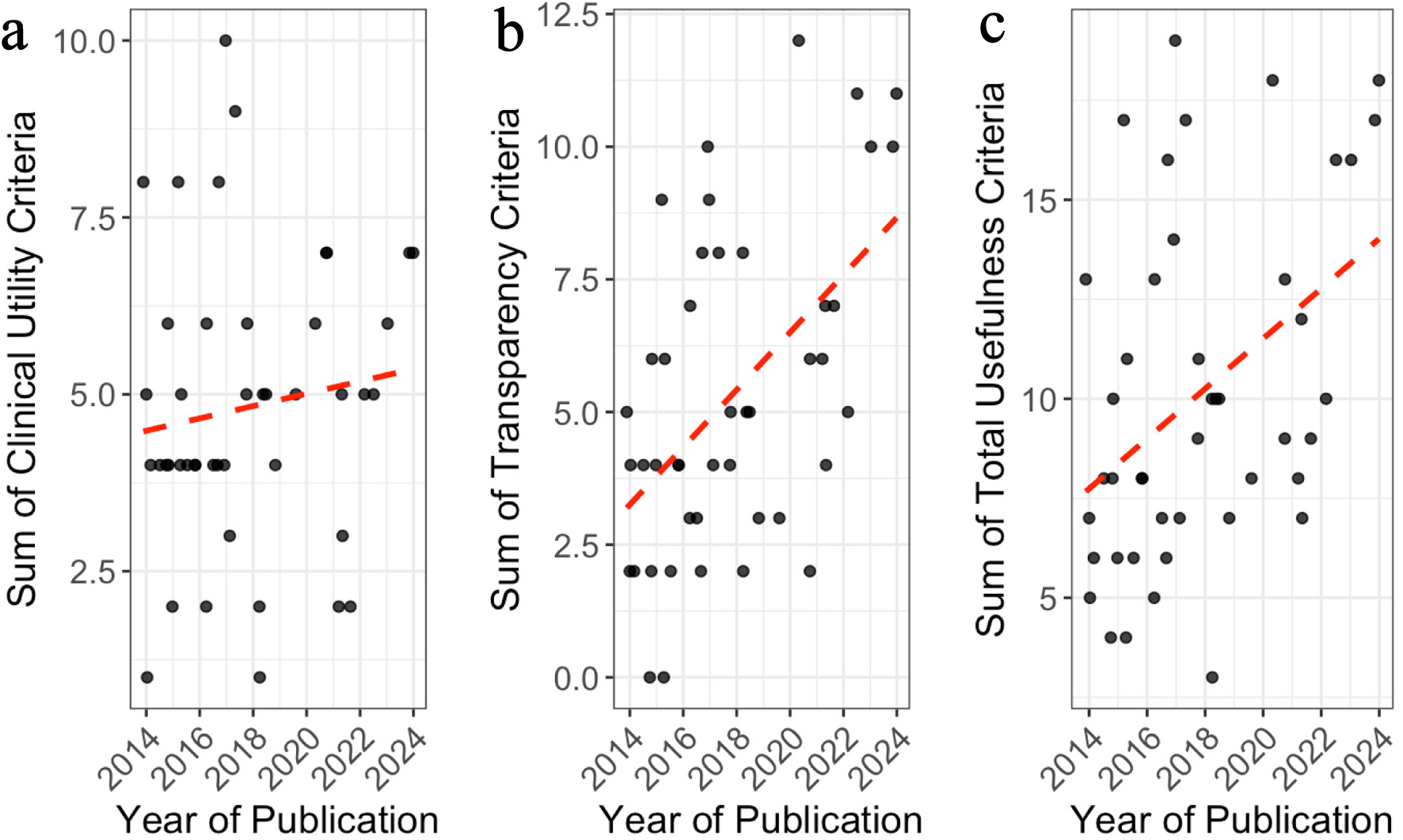
Results: Among 659 records screened, 44 met inclusion criteria. Of these, 15.9% demonstrated information gain, 36.4% provided context placement, and 4.5% avoided violations of pragmatic principles. Patient-centeredness was fully addressed in 54.5% of trials and 38.6% demonstrated feasibility. A majority of studies disclosed funding at 77.3% and conflict of interest statements at 68.2%. Transparency and usefulness values showed a modest upward trend over time (r = 0.42, P < 0.05).
Conclusions: Although CHF RCTs are the gold standard for evaluating new interventions, many fall short in pragmatism, information value, power analysis, and data transparency. Future trials may benefit from prioritizing pragmatic principles, adequate power calculations, cost analysis, and data sharing.
Published
Issue
Section
License
Copyright (c) 2026 The authors

This work is licensed under a Creative Commons Attribution 4.0 International License.









